|
Levamisole (Systemic) VA CLASSIFICATION Primary: IM700 Secondary: AN400 Commonly used brand name(s): Ergamisol. Note: For a listing of dosage forms and brand names by country availability, see Dosage Forms section(s). *Not commercially available in the U.S. Category: Biological response modifier— antineoplastic adjunct— Indications Note: Bracketed information in the Indications section refers to uses that are not included in U.S. Product labeling. Accepted [Carcinoma, colorectal (treatment adjunct)]—Levamisole is indicated, in combination with fluorouracil, for treatment of Dukes C adenocarcinoma of the colon (i.e., with regional lymph node involvement ) after complete resection of primary tumor, with no gross or microscopic evidence of residual disease and no evidence of distant metastases or remaining local metastases that could not be removed en bloc with the primary resection. Why is cocaine adulterated with an animal dewormer? Aminorex, a metabolic of the cocaine adulterant levamisole. Scrotal gangrene after smoking crack. Levamisole slips through this process, meaning you can produce more volume of crack with less pure cocaine. • Levamisole passes the 'bleach test,' a simple street test used to detect impurities in cocaine. Levamisole is a bulking agent for crack. The process of making crack involves. Levamisole passes the. In Surrey addicts are throwing crack pipes. Levamisole is a common cutting agent in crack. The Daily Mirror in England posted a video of a biker making a. It is not useful for therapy of advanced and metastatic disease. Pharmacology/Pharmacokinetics Physicochemical characteristics: Molecular weight— 240.75 Mechanism of action/Effect: Not precisely known. Levamisole appears to act as an immunorestorative agent in the presence of immunosuppression resulting from recent surgery and chemotherapy, but does not stimulate the immune response to above normal levels. May be related to T-cell activation and proliferation, augmentation of monocyte and macrophage activity (including phagocytosis and chemotaxis), and an increase in neutrophil mobility, adherence, and chemotaxis. Does not have cytotoxic effects. Other actions/effects: Anthelmintic. Also has cholinergic, mood-elevating, and, at high doses, convulsant effects. Inhibits alkaline phosphatase in animals. Absorption: Rapidly absorbed from gastrointestinal tract. Biotransformation: Hepatic, extensive. Half-life: Levamisole—3 to 4 hours. Metabolites—16 hours. Time to peak plasma concentration 1.5 to 2 hours. Elimination: Renal, 70% over 3 days (less than 5% unchanged ); fecal, 5% (less than 0.2% unchanged). Precautions to Consider Carcinogenicity Adequate studies in animals have not been done. Studies at doses of 5, 20, and 80 mg per kg of body weight (mg/kg) per day for up to 18 months in mice and up to 24 months in rats found no evidence of carcinogenicity; however, these studies were not conducted at the maximum tolerated dose, and there is a possibility that the animals may not have been exposed to a reasonable drug challenge. Chronic administration of high doses (25 mg/kg) in New Zealand Black mice increased the rate and intensity of spontaneous lymphomas. No carcinogenic effect was found in 12- to 18-month studies in dogs. Mutagenicity Levamisole was not found to be mutagenic in dominant lethal studies in male and female mice, in an Ames test, and in a study to detect chromosomal aberrations in cultured peripheral human lymphocytes. Pregnancy/Reproduction Fertility— Administration through 3 generations of rats and rabbits did not affect fertility. No adverse effects on male or female fertility were noted in rats given oral doses of 2.5, 10, 40, and 160 mg/kg. In a rat gavage study at doses of 20, 60, and 180 mg/kg, the copulation period was increased, the duration of pregnancy was slightly increased, and fertility, pup viability and weight, lactation index, and number of fetuses were decreased at a dose of 60 mg/kg. No adverse reproductive effects occurred when the offspring were allowed to mate and litter. Pregnancy— Adequate and well-controlled studies in humans have not been done. Studies in rats and rabbits at oral doses up to 180 mg/kg found no evidence of fetal malformations. Embryotoxicity occurred at doses of 160 mg/kg in rats and was significant in rabbits at doses of 180 mg/kg. FDA Pregnancy Category C. Breast-feeding It is not known whether levamisole is distributed into human breast milk; however, it is distributed into cows' milk. 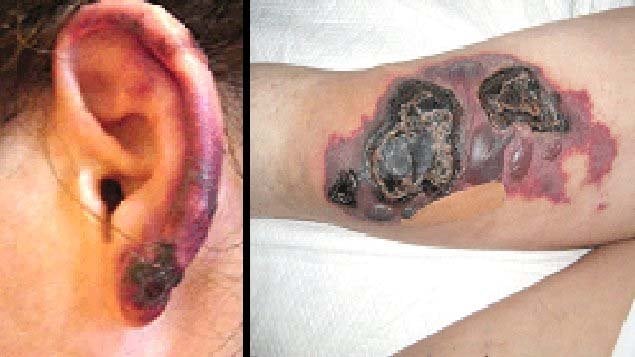
Making Crack With CocainePediatrics No information is available on the relationship of age to the effects of levamisole in pediatric patients. Safety and efficacy have not been established. Geriatrics Appropriate studies on the relationship of age to the effects of levamisole have not been performed in the geriatric population.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2019
Categories |
 RSS Feed
RSS Feed
